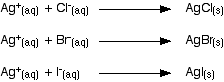
However, the fastest reaction is with an iodoalkane. The electronegativity difference between carbon and iodine is negligible. That means that there will be more attraction between a lone pair on the water and a carbon atom attached to a chlorine atom than if it was attached to an iodine atom. 
The slight positive charge on the carbon will be larger if it is attached to a chlorine atom than to an iodine atom. It is tempting to think that the reaction will be faster if the electronegativity difference is greater. It is slightly positive because most of the halogens are more electronegative than carbon, and so pull electrons away from the carbon. If you have looked at the mechanisms for these reactions, you will know that a lone pair on a water molecule attacks the slightly positive carbon atom attached to the halogen. In order for a halide ion to be produced, the carbon-halogen bond has to be broken. The carbon-iodine bond is the weakest and the carbon-chlorine the strongest of the three bonds. The order of reactivity reflects the strengths of the carbon-halogen bonds. A primary chloro compound probably won't give any precipitate until well after you have lost interest in the whole thing!.A primary bromo compound takes longer to give a precipitate.A primary iodo compound produces a precipitate quite quickly.But the pattern of results is always the same. Obviously, the time taken for a precipitate of silver halide to appear will depend on how much of everything you use and the temperature at which the reaction is carried out. 
You might, for example, compare the times taken to produce a precipitate from this series of primary halogenoalkanes: You would have to keep the type of halogenoalkane (primary, secondary or tertiary) constant, but vary the halogen. Secondary halogenoalkanes do a bit of both of these.Ĭomparing the reaction rates as you change the halogen However, you might be asked to suggest a reason why Fluoride doesn't form a ppt at all - Fluoride ions are so small and lack much shielding of their outer electrons, making it unlikely to become involved in any reaction as this would involve the formation of new bonds which requires a new distribution of electrons.\( \newcommand\] You will not be asked for an explanation of why or how some ppts redissolve and others do not. Note that the Sodium and Nitrate ions are simply spectators and have been omitted. Write an ionic equation to show the formation of a precipitate when aqueous Silver Nitrate is adding to a solution of Sodium Bromide. Remember that only the ions actively involved in forming the precipitate should be shown, and that precipitates are solids.Įg. The examiner may ask for ionic equations containing state symbols Italic text. And the Bromide precipitate can seem white and be confused with the Chloride.įor this reason, at AS you must know that only a white ppt that redissolves when dilute ammonia is added is definitely proof of the Chloride ion.Īnd a yellow precipitate that does not redissolve even with the most concentrated ammonia solution is definitely Iodide. In practice, the Chloride precipitate is always white, but the Iodide precipitate may not be very yellow and could be confused with the Bromide. In reality, a solution of any Silver compound would work since it is the Ag + ions that bond to the halide ions forming insoluble precipitates.Īs in the photo above, Fluoride ions do not form a precipitate at all, Chloride forms a white precipitate, Bromide forms a cream precipitate, and Iodide forms a yellow precipitate. It still involves adding aqueous Silver Nitrate and looking for a precipitate. From the left: Iodide, Bromide, Chloride and Fluoride ions with Silver Nitrate solution
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
